The United States settled claims brought by our client against Massachusetts company Study Across the Pond LLC (SATP) and its principal, John Borhaug. Under the terms of the agreement, SATP will pay $1.3 million to resolve allegations that it violated the False Claims Act by knowingly causing foreign schools in the United Kingdom (UK) to submit false claims and false statements to the U.S. Department of Education in connection with the Direct Loan Program through arrangements that violated the federal ban on incentive-based compensation.
The conduct was brought to light by a qui tam complaint filed by our client. Pursuant to the FCA, our client will receive a relator share of $240,500, plus reasonable attorneys’ fees and costs.
Our client filed the qui tam complaint in 2021. The case is captioned United States ex rel. Hitrost, LLC v. Study Across the Pond, LLC, et al., No. 21-CV-10274-ADB (D. Mass.)
The Complaint alleged that SATP was violating the Incentive Compensation Ban by signing recruitment contracts with U.K. universities that provided for per-student payments for U.S. students recruited to attend these schools.
The Incentive Compensation Ban conditions higher education institutions’ participation in federal student aid programs, upon their agreement not to provide any commission, bonus, or other incentive payment to student recruiters based directly or indirectly on success in securing student enrollments. The Incentive Compensation Ban protects students against aggressive recruitment practices that serve the financial interest of the recruiter rather than the educational needs of the student.
After several years of investigation, the United States intervened in our client’s case. The United States alleged that SATP’s scheme involved at least 28 schools in the UK. Specifically, the United States alleged that SATP demanded a commission for its recruitment services. In many cases, this was money the schools had claimed from the Direct Loan Program for the education of American students. The United States further alleged that SATP created sham records to hide these tuition-sharing arrangements from the Department of Education and ultimately caused foreign schools to submit false claims to the Direct Loan Program.
The Incentive Compensation Ban has historically been a difficult to enforce area of government priority. The United States has rarely intervened in these matters. Similarly, very few – if any – cases have been brought related to American students studying abroad.
This unique case has had an impact that reaches beyond the United States. It has also helped to facilitate a national conversation in the U.K. around student recruitment practices.
The government’s press release emphasized the importance of protecting students from individuals and entities placing their profits ahead of the student’s best interest.
My office is committed to ensuring American students are not taken advantage of for financial gain and protecting the integrity of federal student financial aid programs.
U.S. Attorney Leah B. Foley for the District of Massachusetts
American students deserve to make enrollment decisions free of the improper influence of third-party recruiters who pursue their own financial gain rather than the students’ best interests.
Assistant Attorney General Brett A. Shumate of the Justice Department’s Civil Division
FCA investigations and litigations require coordination among many government agencies and personnel. Here, they came together to ensure that the federal student loan programs are protected from profiteers.
Today’s settlement is a result of the hard work and effort of the Office of Inspector General, the U.S Department of Education, and the U.S. Department of Justice to protect and maintain the integrity of the Federal student aid programs by enforcing applicable laws, including the incentive compensation ban.
Jason Williams, Assistant Inspector General for Investigation Services, U.S. Department of Education Office of Inspector General
Whistleblower Law Collaborative commends the outstanding efforts of their client and the government prosecutors.
We are especially grateful for our client who shed light on this problematic practice and for the government team that doggedly investigated and litigated this matter. AUSAs Alexandra Brazier and Brian LaMacchia, along with DOJ Civil Fraud Trial Attorney Allison Carroll committed countless hours to this investigation and litigation. They refused to ignore the alleged conduct. We are grateful to each of these dedicated public servants.
Erica Blachman Hitchings, WLC Partner
WLC is also grateful to its co-counsel, Whistleblower Partners and Constantine Cannon, for identifying this important matter and their collaboration throughout the life of the case.
WLC has been very active in the Medicare Advantage space. In one instance, we worked with two clients to file a qui tam lawsuit under the False Claims Act. The case, filed in 2020, is captioned United States ex rel. Butler, et al., v. Shikara, et al., No. 20-cv-80483 (S.D. Fl.) and included several Medicare Advantage insurers, as well as an insurance brokerage, provider group, and a physician, as defendants.
Our clients both worked at the defendant insurance brokerage, Well-Life Group, LLC. Though ostensibly owned by his wife, our clients alleged that the brokerage was controlled by Dr. Mazin Shikara, who himself owned and controlled a large primary care practice and management services organization. Based on their experience at the brokerage, Butler and Philipp alleged that several Medicare Advantage insurers knowingly paid Dr. Shikara and his organizations to enroll patients in their Medicare Advantage plans. Our clients asserted that such conduct violated the Anti-Kickback Statute and led to the submission of tainted (and thus, false) claims to Medicare for enrollees.
As WLC partner Erica Blachman Hitchings notes:
It was a true privilege to work with two clients who were willing to alert the government to misconduct that targets the ever-growing number of seniors opting for Medicare Advantage plans.
This is particularly true because these clients came forward in 2020, well before the U.S. Department of Health and Human Services issued a Special Fraud Alert warning about the dangers of kickbacks to brokers in exchange for referrals in the Medicare Advantage marketplace. The allegations in this case fall squarely within the concerns that HHS-OIG identified in its 2024 Special Fraud Alert.
A Medicare beneficiary often relies on — and trusts — their insurance agent to direct them to insurance plans and providers that best fit their needs. When, as alleged here, an agent’s recommendation is “skewed” by an illegal kickback, Medicare enrollees can face “significant and detrimental consequences.”
We take great pride in not only turning out great legal work product, but in helping our clients understand and level-set through each step of a long journey. Reflecting on the experience, client Brian Butler noted:
Erica demonstrated exceptional expertise—particularly regarding Medicare Advantage Plans (‘Part C’)—and a depth of knowledge in this area that stood apart from most other attorneys I consulted. She was consistently accessible throughout the process and took the time to walk us through each step of the case, explaining complex issues in a clear and reassuring way. Her initial filing of the complaint laid critical groundwork that ultimately contributed to a successful outcome.
While WLC is proud to have represented Butler and Philipp from “Day 1,” the entire process has been a team effort. In addition to an early collaboration with McCabe Rabin, WLC was grateful to work hand-in-hand with Amy Easton and Jeffrey Dickstein of Phillips & Cohen LLP, as well as Scott Terry and Chris Gray of Florin Gray.
Notably, Florin Gray handled the litigation following the government’s decision to not intervene in the case. This included an intensive period of discovery, as well as successfully fending off several motions to dismiss and a motion for summary judgment.
WLC attorney Erica Blachman Hitchings notes:
The Florin Gray team dove right into the case, working tirelessly for the next two years to achieve this resolution. It was a true privilege to collaborate with Scott and Chris. Their litigation prowess, endless work ethic, and deep commitment to the clients was evident day in and day out.
In addition, we appreciate the continued engagement of the Department of Justice, which monitored the litigation, filed two statements of interest, and reviewed the settlement before consenting to dismissal of the claims.
The matter resolved before trial, with no determination of liability.
A client of Whistleblower Law Collaborative co-founders Robert M. Thomas, Jr. and Suzanne E. Durrell along with Hirst Law Group, P.C. and Bird, Marella, Rhow, Lincenberg, Drooks, & Nessim, LLP reached a settlement in 2024 of over $47.3 million in a whistleblower case filed in 2015 against The Ensign Group, Inc., and Ensign Services, Inc. (“Ensign”). The settlement resolved the whistleblower client’s allegations that Ensign, who operates skilled nursing facilities nationwide, knowingly engaged in fraud perpetrated against Medicare, Medicaid, and other government health care programs. Ensign acknowledged the settlement at page 65 of its 2024 Annual Report. The claims resolved by the settlement are allegations only and there has been no determination of civil liability.
The complaint, filed as a qui tam suit under the federal and California False Claims Acts (FCAs), alleged that nationwide Ensign knowingly paid kickbacks to physicians for referrals of patients to Ensign’s skilled nursing facilities (SNFs) in violation of the FCAs, the federal and California Anti-Kickback Statutes, and the federal Stark self-referral law. The complaint also alleged that Ensign violated a previous Corporate Integrity Agreement (CIA) that Ensign signed with the Department of Health and Human Services (HHS) in 2013 as part of an earlier False Claims Act case settled by the company.
The whistleblower was a former Contracts Manager at Ensign. In that role, she was responsible for reviewing, tracking, and monitoring contracts entered into by Ensign’s Facilities. She also served on the Company’s Compliance Committee where one of her responsibilities was to ensure that payment provisions in Ensign’s contracts were in accordance with the law.
The whistleblower’s Second Amended Complaint alleged that, as part of her work, she observed, among other fraud, that:
The whistleblower alleged that she complained about the fraud she observed but could not get Ensign to change its conduct. In particular, she alleged that she tried to change the contracts to institute hourly rates tied to actual work and fair market value, rather than excessive monthly lump sum payments to induce referrals, but her attempts were overridden by Ensign.
Patients deserve to know that their doctors’ recommendations for services and the place of service are not tainted by illegal kickbacks. Unfortunately, kickbacks can undermine competition, lead to medically unnecessary services, and drive up the cost of health care.
–Suzanne E. Durrell, Whistleblower Law Collaborative
When the government declined to intervene to litigate the FCA case in 2020, but allowed the whistleblower to proceed, her attorneys at Whistleblower Law Collaborative and Hirst Law Group went forward with the case with Bird Marella as taint counsel.
Sometimes the government declines to intervene in an FCA case for reasons unrelated to the merits of the case; we are glad the government trusted us to move the case forward and that we were successful.
–Robert M. Thomas, Jr., Whistleblower Law Collaborative
The team proceeded to successfully litigate the case for four years and were in the midst of discovery when the settlement was reached in 2024. Gary Lincenberg of Bird Marella applauded the client for her willpower in seeing through this lengthy matter which was difficult to relive and thanked his co-counsel for their excellent work.
We congratulate the whistleblower for her courage and persistence. She has shown that one person, willing to stand up for what she knows is right, can make a difference. She is exactly the kind of deserving person that the False Claims Act envisions. Without her courage in coming forward, none of this would likely have come to light.
–Michael A. Hirst, Hirst Law Group
For her efforts in bringing and prosecuting the FCA case over nine and one-half years, the government awarded the whistleblower a share of the settlement.
The United States settled a False Claims Act case brought by two of our clients against Genexe, LLC d/b/a Genexe Health, its parent company Immerge, Inc., and two of the companies’ control persons and owners, Jason Green and Jason Gross. Under the terms of the Genexe settlement, Genexe, Immerge, Gross and Green will collectively pay $6 million plus interest to resolve the allegations of genetic testing fraud.
The fraudulent conduct was brought to light by multiple FCA qui tam cases filed by several whistleblowers, including two of our clients. The whistleblowers collectively will receive a relators’ share award of approximately $1.3 million from the settlement. Defendants are also paying the Relators’ reasonable attorneys’ fees and costs.
After investigating the claims raised in the relators’ FCA qui tam complaints, the United States alleged that during 2018 and 2019, Genexe, Immerge, Green, and Gross assisted with and caused to be submitted false claims to Medicare for CGx and PGx tests that were not medically necessary and that were procured through kickbacks. offered and paid to a network of independent contractors (IBOs), medical laboratories, medical providers, and telemedicine healthcare providers. Medicare does not cover clinical laboratory tests that are not medically necessary or that are tainted by kickbacks.
Genexe would obtain the genetic samples (usually from a cheek swab of saliva) for the CGx and PGx tests along with Medicare beneficiaries’ protected health care information and a physician order for the genetic test and send the Medicare beneficiary’s specimen and information to the laboratories for testing. Laboratories then billed Medicare for the fraudulent genetic testing and were regularly reimbursed at rates exceeding $6,000 per test. Once Medicare paid the medical laboratory, Genexe would receive a portion of the Medicare reimbursement funds from the laboratory; initially, Genexe was paid about $800 per swab, but later was paid amounts ranging from $1,000 to $2,000 depending on the type of genetic test.
This settlement is particularly notable because Jason Green and Jason Gross, two individual officers and owners of the companies, were held accountable by the United States and will collectively pay the settlement amount along with the companies. Green and Gross served, respectively, as the Chief Executive Officer and Chief Operating Officer of Genexe and Immerge, and they controlled and had ownership interests in the companies.
In the press release announcing the settlement, government officials emphasized the importance of identifying and ending genetic testing fraud scams.
Genetic testing fraud preys on the fears of patients, and it wastes taxpayer dollars by spending limited funds on medically unnecessary or nonexistent tests. This settlement shows we will work with our law enforcement partners to investigate fraud, waste, and abuse in federal healthcare programs and will use every tool available to recover improperly paid taxpayer funds.
United States Attorney David Metcalf
Medical professionals should only order testing which would benefit individual patient care, not for personal gain.
Maureen Dixon, Special Agent in Charge for the U.S. Department of Health and Human Services
Whistleblower Law Collaborative commends the outstanding efforts of their clients and the government prosecutors.
It was a privilege to represent two clients who were willing to come forward to alert the government to a national fraud scheme which preyed on elderly victims and cost taxpayers millions of dollars. The settlement announced today is a testament to the skill, resolve, and stamina of an outstanding team of government attorneys and investigators who put an end to that scheme and held those who profited accountable for their actions.
Bruce C. Judge, Partner, Whistleblower Law Collaborative
This is the third settlement of a genetic testing fraud FCA case brought by a client of WLC. We began sounding the alarm about genetic testing scams in 2018. Without the courage of whistleblowers, scams like this would go undetected.
MORSECORP, Inc. has agreed to pay $4.6 million, plus interest, to resolve allegations that it made false representations concerning compliance with required cybersecurity controls for safeguarding sensitive government information. This settlement is particularly notable because it represents the first major False Claims Act settlement with a defense contractor based on failures to implement required cybersecurity controls.
MORSECORP d/b/a MORSE (Mission Oriented Rapid Solution Engineering), which is based in Cambridge, Massachusetts, is a defense contractor in the U.S. National Security Ecosystem. MORSE performed multiple contracts for the Department of the Army and the Department of the Air Force, among other government customers.
The case was brought by our client in January 2023 based on his concerns that MORSECORP had not fully implemented cybersecurity controls required under NIST SP 800-171 for protecting sensitive government data and information. Our client was also concerned that MORSECORP did not have a consolidated system security plan and that MORSECORP was using third-party cloud-based services that did not meet the relevant Federal security requirements. In addition, our client was concerned that MORSECORP had posted improperly inflated SPRS assessment scores with the Defense Department for its internal cybersecurity practices and policies.
Our client brought the MORSECORP cybersecurity failures to the attention of the government by filing a cybersecurity qui tam complaint under the False Claims Act. Under the False Claims Act, a private citizen (known as a “relator”) who suspects or knows of fraud against the government can act as a whistleblower and file a sealed complaint on behalf of the government. If the case is successful, the relator is entitled to a share – between 15% and 30% – of the government’s recovery.
The government investigated the allegations brought to light by our client and, on March 17, 2025, filed a notice of its intention to intervene against MORSECORP for the purpose of settlement. The Department of Justice further described the case and the $4.6 million settlement on March 25, 2025.
Federal contractors must fulfill their obligations to protect sensitive government information from cyber threats, We will continue to hold contractors to their commitments to follow cybersecurity standards to ensure that federal agencies and taxpayers get what they paid for, and make sure that contractors who follow the rules are not at a competitive disadvantage.
— United States Attorney Leah B. Foley.
Protecting the integrity of Department of Defense (DoD) procurement activities is a top priority for the DoD Office of Inspector General’s Defense Criminal Investigative Service (DCIS). Failing to comply with DoD contract specifications and cybersecurity requirements puts DoD information and programs at risk. We will continue to work with our law enforcement partners and the Department of Justice to investigate allegations of false claims on DoD contracts.
— Special Agent in Charge Patrick J. Hegarty, DCIS Northeast Field Office
Our client expresses his admiration for, and appreciation of, the outstanding efforts of the Commercial Litigation Branch of the U.S. Department of Justice, the U.S Attorney’s Office for the District of Massachusetts, and the relevant investigating agencies for conducting a thorough and prompt investigation which led to the settlement announced yesterday.
In uniform and out, protecting the national security of the United States has been the focus of my professional career. Becoming a whistleblower was not an easy decision and one I only took when I felt I had no remaining option to protect sensitive government information. The Department of Justice should be commended for acting promptly to investigate and put an end to practices that placed sensitive government information and data at risk of loss or compromise.
–WLC Client
Whistleblower Law Collaborative commends the outstanding efforts of their client and the government prosecutors. Attorney Bruce C. Judge praised his client’s willingness to stand his ground on protecting sensitive government information. Mr. Judge also cited his client’s courage in coming forward without regard to the professional and personal risks that can attach to being a whistleblower. Mr. Judge continued,
[O]ur client drew on his extensive knowledge of the applicable cybersecurity requirements and was able to identify numerous cybersecurity gaps clearly and persuasively to government prosecutors and agents.
This settlement marks an important milestone in the government’s long-running efforts to bring about cybersecurity compliance in the Defense Industrial Base. After many years of issuing warnings and relying on self-certifications, the MORSECORP settlement sends a clear signal that government contractors who fail to implement required cybersecurity controls can expect to face significant financial penalties. It also signals that individuals who bring cybersecurity violations to the government’s attention can receive a share of the government’s ultimate recoveries.
In 2021, the Department of Justice launched its Civil Cyber-Fraud Initiative aimed at federal contractors who fail to comply with government cybersecurity requirements. In addition, the initiative targets contractors who fail to report breaches or other cybersecurity incidents.
We expect the Department of Justice, the Department of Defense, NASA, and other government agencies to continue to investigate and prosecute matters involving failure to implement required cybersecurity controls to safeguard sensitive government information and data.
This settlement among the United States, the States, and pharmaceutical companies Mallinckrodt plc and its subsidiary Mallinckrodt ARD LLC resolved allegations that Mallinckrodt violated the federal and state False Claims Acts by knowingly underpaying Medicaid rebates for its high-priced drug Acthar. The total settlement amount is $233,707,865 (plus interest). The United States will receive $123,642,146, and States will receive $110,065,718.
Mallinckrodt knowingly misreported Acthar’s base Average Manufacturer Price (“base AMP”) from January 2013 through June 2020. By doing so, it reduced the rebates it paid to the Medicaid Drug Rebate Program (MDRP) by approximately $650 million. Mallinckrodt had increased Acthar’s price from approximately $50 per vial in 2001 to almost $40,000 per vial.
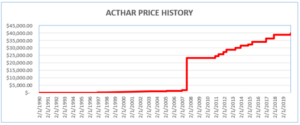
Where a drug’s price is increased above the rate of inflation, manufacturers must pay an additional rebate. To avoid meeting its increased rebate obligations, Mallinckrodt began reporting Acthar’s base AMP as if it had been approved in 2010 (after the enormous price increases). Acthar, however, had been approved in 1952.
Our client, James Landolt, served as Mallinckrodt’s Director of Internal Controls, Gross to Net Accounting and Government Reporting from November 2015 until July 2017. In that position, he learned that Mallinckrodt had been misreporting the base AMP for Acthar and had underpaid the MDRP by hundreds of millions of dollars.
Mr. Landolt resigned from Mallinckrodt in 2017 and filed a qui tam action in 2018 alleging that Mallinckrodt’s knowing failure to pay correct rebates for Acthar violated federal and state False Claims Acts. In March 2020, the United States intervened in his lawsuit. In June 2020, twenty-eight states, the District of Columbia, and Puerto Rico also intervened.
While the False Claims Act was still under seal, Mallinckrodt sued the Center for Medicare and Medicaid Services (CMS) in federal court in the District of Columbia. Mallinckrodt sought a ruling that it was correctly reporting Acthar’s base AMP and did not have to comply with instructions from CMS to correct its reporting and pay what it owed. In March 2020, the District Court rejected Mallinckrodt’s argument. Two months later, it rejected Mallinckrodt’s motion for reconsideration and for a preliminary injunction.
In October 2020, Mallinckrodt filed for bankruptcy, which stayed the pending False Claims Act case. On March 2, 2022, the bankruptcy court confirmed Mallinckrodt’s plan of reorganization, which included this $234 million settlement.
As part of the settlement, Mallinckrodt entered into a five-year Corporate Integrity Agreement with HHS-OIG that requires, among other things, an independent review organization to annually review multiple aspects of the company’s practices relating to the Medicaid Drug Rebate Program. Mallinckrodt began reporting the correct base AMP for Acthar in June 2020 after losing its case in United States Court in D.C.
Mr. Landolt will receive a 20% share of amounts paid under the federal and state False Claims Acts. Whistleblowers like Mr. Landolt are critical in the fight against fraud. Under the False Claims Act, a private citizen-relator who suspects or knows of fraud against the government can file a sealed complaint on behalf of the government. In successful cases, the relator is entitled to a share of the government’s recovery.