May 16, 2023
The Inflation Reduction Act of 2022 (“Act”) contains important Medicare prescription drug provisions. These include:
This fact sheet, prepared by the Centers for Medicare & Medicaid Services, summarizes this important legislation. Here, we focus on the Act’s Medicare drug rebate provisions.
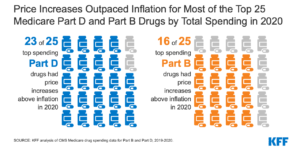
The Medicaid Drug Rebate Program was created in 1990. Up until now, Medicare had no similar program. The Act, however, includes a requirement that drug manufacturers pay rebates if prices for drugs used by Medicare beneficiaries rise faster than inflation. According to an analysis by the non-profit KFF, from 2019 to 2020, price increases exceeded inflation for half of all drugs covered by Medicare. The inflation rate was about 1% for that period. However, among those drugs whose price increases exceeded the rate of inflation, one-third had price increases of 7.5% or more.
Significantly, 23 of the top 25 prescribed Medicare Part D drugs and 16 of the top 25 prescribed Medicare Part B drugs had price increases that outpaced inflation in 2020.
The Act’s Part D inflation rebate provisions took effect in October 2022. The Part B inflation rebate provisions took effect in the first quarter of 2023. Under both, manufacturers that fail to pay the required rebate will face a civil monetary penalty equal to at least 125% of the original rebate amount.
In addition to civil monetary penalties, a drug company that does not pay rebates as required risks liability under the False Claim Act. Several cases have resulted in large judgments against drug companies that evaded their responsibilities under the Medicaid Drug Rebate Program (MDRP). Our firm represented whistleblowers in two major Medicaid rebate fraud cases.
Most recently, we represented the whistleblower in a case against Mallinckrodt, which resulted in a $234 million settlement. That case alleged that Mallinckrodt cheated the MDRP out of hundreds of millions of dollars by misreporting pricing data to avoid paying inflationary rebates. Specifically, our client alleged that Mallinckrodt ARD improperly used a base Average Manufacturer’s Price (“AMP”) from 2010 for its high-priced drug Acthar. However, because Acthar first entered the market in the 1950s, its base AMP should have been calculated from 1990. From 1990 to 2010, Acthar’s price dramatically outpaced inflation. By reporting a later base AMP, Mallinckrodt improperly reduced the additional rebate owed to Medicaid by hundreds of millions of dollars.
In 2017, we successfully represented a client in another Medicaid rebate fraud case. In that case, Mylan paid $465 million for misclassifying EpiPen® as a generic drug to avoid paying the higher rebates owed on brand name drugs.
The Whistleblower Law Collaborative LLC represents clients nationwide in bringing actions under state and federal False Claims Acts. In addition, the firm assists clients in bringing claims in other whistleblower programs. Among the firm’s many successes are the Mallinckrodt and Mylan cases discussed above. For his role in exposing Mallinckrodt’s misconduct, our client received a relator’s share of over $40 million. If you are aware of Medicare or Medicaid drug rebate fraud, please contact us for a free consultation.